Excipient
ICH-Q7 GMP Manufactured ProductMES, Monohydrate, Low PVS
GMP Excipient Grade
Product Code: MESM-3222 | Previously: ME3222
Intended For Use In Pharmaceutical GMP Processes/Excipient
MES Monohydrate is a zwitterionic buffer that is not absorbed through cell membranes and is virtually transparent in UV light. MES is a buffering agent used in many biological and biochemical applications. It is also used as a running buffer for denaturing gel electrophoresis. The characteristics of low UV absorptivity, minimal reactivity, stable pH and solubility in water allow MES Monohydrate Excipient to be used as a Good’s buffer.
Product Specifications
| ANALYSIS | SPECIFICATIONS | |||
|---|---|---|---|---|
|
0.1000 a.u. max. 0.1000 a.u. max. |
|||
|
White Crystalline Powder | |||
|
99.5% min. | |||
|
0.005% max.. | |||
|
Colorless | |||
|
None Detected None Detected None Detected |
|||
|
2 ppm max. | |||
|
Conforms to Reference | |||
| Loss on Drying @ 130°C | 7 - 10% | |||
|
3.1 - 3.5 | |||
|
2.7 – 3.7 | |||
|
2.5 – 4.5 | |||
|
5.9 – 6.3 |
|||
|
≤ 1 ppm | |||
|
0.05% max. | |||
|
Passes Test | |||
|
0.005% max. | |||
|
2 ppm max. 2 ppm max. 2 ppm max. 2 ppm max. |
|||
|
7.9 – 8.9% |
COAs and Tech Docs
- Printable Spec Sheet
- Product Inquiry
- C of As
- SDS
- Regulatory Packet
- Process Flow Diagram
- GMP Statement
- Animal Origin Statement
- BSE/TSE
- Allergen
- Mycotoxin/Aflatoxin
- GMO Statement
- Melamine
- Residual Solvents
- Gluten Free
- Ingredient Declaration
- REACH
- Prop 65
- Supply Chain
- Imidazole
- Formaldehyde
- Slip Agent Statement
- Genotoxic Impurities
- Phthalate, Bisphenol, Dioxin
- Catalyst
- Test Methods
- IRGAFOS
- Degradation and Impurity
- Nitrosamine Risk Assessment
- Elemental Impurity Assessment
- Label Copy
- Certificate of Origin
- Latex Statement
- Real-Time Stability Report
- TUPP Report- Bangor

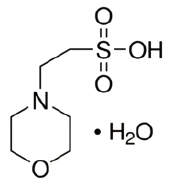
MESM-3222
CAS #: 145224-94-8
Formula: C6H13NO4S·H2O
Sol. In H2O (g/L): 310
F.W.: 213.25 g/mol
pH @ 20°C: 2.5 - 4.5
Useful pH: 5.5 - 6.7
pKa @ 20˚C: 6.1
General Product Description:
- The manufacturing of Bio Excipient Grade MES, Monohydrate MESM-3222 is performed at BioSpectra’s Bangor, PA facility utilizing multiuse equipment. Equipment used in the manufacturing of Bio Excipient Grade MES, Monohydrate is cleaned in accordance with
- BioSpectra’s Cleaning Validation Plan.
- MES is a White Crystalline product.
- Molecular Formula: C6H13NO4S·H2O
- Molecular Weight: 213.25 g/mol.
- CAS Number: 145224-94-8.
- There are no known major food allergens (as defined by FDA and WHO) in the manufacture of this product.
- BioSpectra certifies that all MES, Monohydrate MESM-3222 manufactured at BioSpectra and its raw materials are not derived from or come in contact with animal parts, products, and/or byproducts.
- MES, Monohydrate manufactured at BioSpectra and any raw materials used in the manufacture of MES, Monohydrate at BioSpectra are not subject to genetic modification.
- Synonyms: 2-(N-Morpholino)ethanesulfonic acid, 4-Morpholineethanesulfonic acid monohydrate.
GMP Compliance:
Bio Excipient Grade MES, Monohydrate MESM-3222 is suitable for use as an excipient. It is manufactured in accordance with the ICH-Q7 Good Manufacturing Practice Guide. This grade of MES, Monohydrate is not suitable to be used as an Active Pharmaceutical Ingredient, Drug Product or Household Item.
Retest Date:
The recommended retest period for MES, Monohydrate is two years from the date of manufacture.
Storage and Shipping Conditions:
Ship and Store in ambient temperature.
Package Sizes:
10kg, 25kg and 50kg pails.
Additional Packaging Information
https://www.biospectra.us/technical/packaging


