Excipient
ICH-Q7 GMP Manufactured ProductSODIUM DECANOATE
Blend Batch, GMP Excipient Grade
Product Code: NDEC-3320 | Previously: ND3320
Intended For Use As An Excipient
Sodium Decanoate – Blend Batch, is a the product code for the blend of multiple production batches to provide the end-user with a larger blended, uniform Lot size. Sodium Decanoate, or sodium caprate, is the sodium salt of caproic acid, a 10-carbon saturated fatty acid. It has amphiphilic character and can form micelles and liquid crystalline phases in aqueous solution. Sodium decanoate’s properties may help elucidate the transport of biologically active molecules and as a final component of a finished drug product, it may serve to enhance the bioavailability of the API. BioSpectra’s Bio Excipient Grade of Sodium Decanoate is supported by a Type IV drug master file submitted with the FDA.
Product Specifications
|
ANALYSIS |
SPECIFICATIONS |
||
|---|---|---|---|
|
White to off-white Powder | ||
|
97.0% – 103.0% | ||
|
Passes Test | ||
|
3.0% max. |
||
|
9.0 – 11.0 |
||
|
<1.0% | ||
|
Passes Test | ||
|
Passes Test | ||
|
1.5% – 3.0% |
COAs and Tech Docs
- Printable Spec Sheet
- Product Inquiry
- C of As
- SDS
- Elemental Impurities
- Regulatory Packet
- Process Flow Diagram
- GMP Statement
- Animal Origin Statement
- BSE/TSE
- Allergen
- Mycotoxin/Aflatoxin
- GMO Statement
- Melamine
- Residual Solvents
- Gluten Free
- Ingredient Declaration
- Prop 65
- Supply Chain
- Imidazole
- TUPP Report - Stroudsburg
- Formaldehyde
- Slip Agent Statement
- Genotoxic Impurities
- Phthalate, Bisphenol, Dioxin
- Catalyst
- Test Methods
- IRGAFOS
- Degradation and Impurity
- Stability Report
- Nitrosamine Risk Assessment
- Label Copy
- Certificate of Origin
- Impurity Analysis Summary
- Analytical Method Validation Report
- Analytical Method of Analysis - Determination of ICH Q3D Elemental Impurities

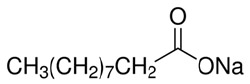
NDEC-3320
CAS #: 1002-62-6
Formula: C10H19NaO2
Sol. In H2O (g/L): 0.1
F.W.: 194.25 g/mol.
General Product Description:
- The manufacturing of Sodium Decanoate NDEC-3320 is performed at BioSpectra’s Bangor, PA facility utilizing multiuse equipment.
- Sodium Decanoate is a White to off-white powder
- Molecular Formula: C10H19NaO2
- Molecular Weight: 194.25 g/mol.
- CAS Number: 1002-62-6
- There are no known major food allergens (as defined by FDA and WHO) in the manufacture of this product.
- BioSpectra certifies that all Sodium Decanoate NDEC-3320 manufactured at BioSpectra and its raw materials are not derived from nor come in contact with animal parts, products and/or byproducts.
- Sodium Decanoate manufactured at BioSpectra and any raw materials used in the manufacture of Sodium Decanoate at BioSpectra are not subject to genetic modification.
- Synonyms: Sodium Caprate, Decanoic acid Sodium salt, Capric acid Sodium salt
GMP Compliance:
Bio Excipient Grade Sodium Decanoate NDEC-3320 is suitable for use as an excipient. It is manufactured in accordance with the ICH-Q7 Good Manufacturing Practice Guide. This grade of Sodium Decanoate is not suitable to be used as an Active Pharmaceutical Ingredient, Drug Product or Household Item.
Retest Date:
The recommended retest period for Sodium Decanoate is two years from the date of manufacture.
Storage and Shipping Conditions:
Ship and Store between 2°C and 8°C. Store in a clean and dry area. Store in the original container.
Package Sizes:
1 kg bottle, 5 kg, 10 kg and 25 Kg pails
Additional Packaging Information
https://www.biospectra.us/technical/packaging


