Buffer/ Excipient
ICH-Q7 GMP Manufactured ProductGUANIDINE HCl Buffer,
NF Grade, GMP Excipient Grade
Product Code: GHCL-3220 | Previously: GH3220
Intended For Critical Biopharma Applications
Guanidine Hydrochloride Buffer is a strong protein denaturant that functions as a chaotropic agent. As a denaturant, it acts to unfold proteins and turn them into their original polypeptide chains. As a chaotropic agent, it breaks down the structure of proteins. Guanidine Hydrochloride Buffer is commonly used in the purification of RNA by dissociating the RNA into its nucleic acids and protein forms. At higher concentrations, Guanidine Hydrochloride Buffer decreases enzyme activity. It is also used to increase the solubility of hydrophobic molecules.
Product Specifications
| ANALYSIS | SPECIFICATIONS | ||
|---|---|---|---|
|
≤0.01% |
||
|
White / Crystals | ||
|
99.5% - 101.0% | ||
| Chloride and Sulfate, Sulfate |
≤0.005% |
||
|
None Detected None Detected None Detected |
||
|
Passes Test | ||
|
≤ 0.2000 a.u. ≤ 0.0300 a.u. ≤ 0.0300 a.u. |
||
|
Identification C – Chloride |
Meets the Requirement of Test A |
||
|
Limit of Nitrate |
≤0.005% | ||
| Loss on Drying | ≤0.5% | ||
|
184ºC-188ºC |
||
|
4.5 – 6.0 | ||
|
≤0.05% | ||
|
Passes Test | ||
|
≤ 5 ppm ≤ 5 ppm ≤ 5 ppm ≤ 5 ppm |
||
|
Water by Karl Fischer |
NMT 0.3%w/w | ||
|
Water Insoluble |
≤0.05% |
COAs and Tech Docs
- Printable Spec Sheet
- Product Inquiry
- C of As
- SDS
- Elemental Impurities
- Regulatory Packet
- Process Flow Diagram
- GMP Statement
- Animal Origin Statement
- BSE/TSE
- Allergen
- Mycotoxin/Aflatoxin
- GMO Statement
- Melamine
- Residual Solvents
- Gluten Free
- Ingredient Declaration
- REACH
- Prop 65
- Supply Chain
- TUPP Report - Stroudsburg
- Formaldehyde
- Slip Agent Statement
- Genotoxic Impurities
- Phthalate, Bisphenol, Dioxin
- Catalyst
- Test Methods
- IRGAFOS
- Stability Report
- Nitrosamine Risk Assessment
- Label Copy
- Certificate of Origin
- Kosher / Halal Statement

GHCL-3220
CAS #: 50-01-1
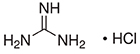
Formula: CH5N3 • HCl
Sol. In H2O (g/L): 573
F.W.: 95.53 g/mol
pH @ 20°C: 4.5 - 6.0
General Product Description:
- The manufacturing of Guanidine Hydrochloride Buffer GHCL-3220 is performed at BioSpectra’s Stroudsburg, PA facility and is conducted in a dedicated processing area using only dedicated equipment.
- Guanidine HCl Buffer is a White Crystalline product.
- Molecular Formula: NH2C(NH)NH2HCl.
- Molecular Weight: 95.53 g/mol.
- CAS Number: 50-01-1.
- There are no known major food allergens (as defined by FDA and WHO) in the manufacture of this product.
- BioSpectra certifies that all Guanidine Hydrochloride Buffer GHCL-3220 manufactured at BioSpectra and its raw materials are not derived from or come in contact with animal parts, products, and/or byproducts.
- Guanidine Hydrochloride Buffer manufactured at BioSpectra and any raw materials used in the manufacture of Guanidine Hydrochloride Buffer at BioSpectra are not subject to genetic modification.
- Synonyms: Guanidine Monohydrochloride, Guanidinium Chloride, Guanidinium Hydrochloride Buffer.
GMP Compliance:
Bio Excipient Grade Guanidine Hydrochloride Buffer, GHCL-3220 is suitable for use as an excipient. It is manufactured in accordance with the ICH-Q7 Good Manufacturing Practice Guide. This grade of Guanidine Hydrochloride Buffer is not suitable to be used as an Active Pharmaceutical Ingredient, Drug Product or Household Item.
Retest Date:
The recommended retest period for Guanidine
Hydrochloride Buffer is two years from the date of manufacture.
Storage and Shipping Conditions:
Ship and Store in ambient temperature.
Package Sizes:
10kg, 25 kg and 50 kg pails.
Additional Packaging Information
https://www.biospectra.us/technical/packaging


