Reagent / Compendial
ICH-Q7 GMP Manufactured ProductTRIS Hydrochloride (HCL)
99.9% High Purity, LBLE, LEI, Reagent GMP Grade
Low Bioburden, Low Endotoxin, Multicompendial, GMP Manufactured,
Reagent Grade Product
Product Code: THCL-3257 | Previously: TH3257
Intended For Use As A High Purity Reagent Grade
For Critical Analytical, Diagnostic and Pharmaceutical Applications, and downstream manufacture of drug products where a high purity, high assay, GMP Tris Hydrochloride is required. Tris Hydrochloride is a considered a “Good’s” buffer because it has low UV absorptivity, minimum reactivity, stable pH and is soluble in water.
Product Specifications
|
ANALYSIS |
SPECIFICATIONS |
||
|---|---|---|---|
|
≤ 0.06 a.u. ≤ 0.06 a.u. ≤ 0.01 a.u. |
||
|
White, Crystalline Powder to Needle-Like Crystals | ||
|
≥ 99.0% | ||
| Bioburden | ≤ 100 CFU/g | ||
| Endotoxin | ≤ 2.5 EU/g | ||
|
None None None |
||
|
Passes Test Passes Test |
||
|
< 0.005% | ||
|
≤ 0.5% | ||
|
147–152ºC | ||
|
4.0 - 5.0 3.5 - 5.0 |
||
|
≤ 5 ppm |
||
|
Water |
≤ 1.0% |
| List of C of As files |
|---|
| No files available right now. |
General Product Description:
- The manufacturing of Tris Hydrochloride THCL-3257 is performed at BioSpectra’s Stroudsburg, PA facility and is conducted in a dedicated processing area using only dedicated equipment.
- Tris Hydrochloride (HCl) Buffer is a White Crystalline product
- Molecular Formula: C4H11NO3 • HCI
- Molecular Weight: 157.60 g/mol.
- CAS Number: 1185-53-1
- There are no known major food allergens (as defined by FDA and WHO) in the manufacture of this product.
- BioSpectra certifies that all Tris Hydrochloride (HCl) Buffer THCL-3257 manufactured at BioSpectra and its raw materials are not derived from or come in contact with animal parts, products and/or byproducts.
- Tris Hydrochloride (HCl) Buffer manufactured at BioSpectra and any raw materials used in the manufacture of Tris Hydrochloride at BioSpectra are not subject to genetic modification.
- Synonyms: Tris Hydrochloride (HCl), 2-Amino-2-(Hydroxymethyl)- 1,3-Propanediol Hydrochloride; Tris (Hydroxymethyl) Aminomethane Hydrochloride
Product Statements:
Plant or Animal Origin Statement: BioSpectra certifies that Tris, Product Code THCL-3257, manufactured by BioSpectra is of Synthetic origin. BioSpectra certifies that Tris, Product Code THCL-3257, is not derived from Plant or Animal Origin.
Metal Residue Statement: BioSpectra certifies that Tris, Product Code THCL-3257, manufactured by BioSpectra is manufactured without the use of metal catalysts and metal reagents. There are no known risks to metal residues in this product.
Melamine Statement: Based on the manufacturing process and the controlled handling, storage, and analysis of Tris, Product Code THCL-3257, BioSpectra certifies that there is low risk for melamine contamination. Additionally, when tested for melamine, this product reports less than 0.15 ppm.
Residual Solvents Statement: Based on the manufacturing process and the controlled handling, storage and analysis of this product, this product complies with the requirements and specifications listed in the current USP method <467> Tables 1, 2, 3, or 4.
GMP Compliance:
Bio Excipient Grade Tris Hydrochloride THCL-3257 is suitable for use as an excipient. It is manufactured in accordance with the ICH-Q7 Good Manufacturing Practice Guide. This grade of Tris Hydrochloride is not suitable to be used as an Active Pharmaceutical Ingredient, Drug Product or Household Item.
Expiration:
The recommended expiration period for Tromethamine Hydrochloride is three years from the date of manufacture..
Storage and Shipping Conditions:
Ship and store in ambient temperature. Store in a clean and dry area. Store in the original container.
Package Sizes:
10kg, 25kg and 50kg drums.
Additional Packaging Information
https://www.biospectra.us/technical/packaging
COAs and Tech Docs
- Printable Spec Sheet
- Product Inquiry
- C of As
- SDS
- TUPP Report - Stroudsburg
- Test Methods
- Elemental Impurity Assessment
- Regulatory Packet – Stroudsburg
- Regulatory Packet – Bangor
- Allergen Statement Bangor
- Allergen Statement Stroudsburg
- Animal Origin Statement Bangor
- Animal Origin Statement Stroudsburg
- BSE / TSE Statement Bangor
- BSE / TSE Statement Stroudsburg
- Catalysts Statement Bangor
- Catalysts Statement Stroudsburg
- Certificate of Origin Statement Bangor
- Certificate of Origin Statement Stroudsburg
- Formaldehyde Statement Bangor
- Formaldehyde Statement Stroudsburg
- Genotoxic Impurity Statement Bangor
- Genotoxic Impurity Statement Stroudsburg
- Gluten Free Statement Bangor
- Gluten Free Statement Stroudsburg
- GMO Statement Bangor
- GMO Statement Stroudsburg
- GMP Statement Bangor
- GMP Statement Stroudsburg
- Imidazole Statement Bangor
- Imidazole Statement Stroudsburg
- Ingredient Declaration Bangor
- Ingredient Declaration Stroudsburg
- IRGAFOS Bangor
- IRGAFOS Stroudsburg
- Melamine Bangor
- Melamine Stroudsburg
- Mycotoxin-Aflatoxin Statement Bangor
- Mycotoxin-Aflatoxin Statement Stroudsburg
- Nitrosamine Risk Assessment Bangor
- Nitrosamine Risk Assessment Stroudsburg
- Phthalate, Bisphenol, Dioxin Bangor
- Phthalate, Bisphenol, Dioxin Stroudsburg
- Process Flow Diagram Bangor
- Process Flow Diagram Stroudsburg
- Prop 65 Bangor
- Prop 65 Stroudsburg
- REACH Bangor
- REACH Stroudsburg
- Residual Solvents Bangor
- Residual Solvents Stroudsburg
- Slip Agent Statement Bangor
- Slip Agent Statement Stroudsburg
- Supply Chain Statement Bangor
- Supply Chain Statement Stroudsburg
- Compliance Statement Bangor
- Compliance Statement Stroudsburg
- TUPP Bangor
- M11 TUPP Report Bangor
- Elemental Impurity Assessment Bangor (M11)
- Elemental Impurity Assessment: S03 Process Validation 2023

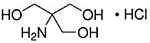
THCL-3257 | TH3257
CAS #: 1185-53-1
Formula: C4H11NO3·HCl
Sol. In H2O (g/L): 1,010
F.W.: 157.60 g/mol
pH @ 20°C: 4.5 - 6.0
Useful pH: 7.0-9.0
pKa @ 20˚C: 8.2


